Graphing in Science Activity
Today you will learn how important experiments and graphing are to science exploration. Below you will find three experiments to test various topics. You will complete the experiment and then graph the results. Lastly, you will read and answer the questions for the document "Graphing in Science".
Experiment One: Tennis Ball Drop
You will drop a tennis ball from 5 heights measuring how far the ball bounces back. Drop the tennis ball from 20cm, 40cm, 60cm, 80cm & 100cm. Each time measuring the distance the ball bounces back up. Before you start, predict which height will causes the greatest bounce and write your prediction in a hypothesis. After you have written your hypothesis you may begin testing each bounce height. After you have completed the experiment you will create a graph showing the relationship between distance dropped and bounce.
Experiment Two: Paper Towel Absorbency
You will test the absorbency (amount of water something can absorb) of three types of paper towels to see which is best at soaking up water. Before you being, examine each paper towel for its characteristics and quality. Make a hypothesis about which paper towel will absorb the most water and why. First, weigh each strip of paper towel and write down the initial mass. Next, dip the paper towel into the water allowing it to become totally wet. Then measure it again to see how much water it absorbed. Compare the three types of paper towel and create a graph to show how much water was absorbed.
Experiment Three: Water Temperature
In this experiment you will see how water changes temperature as it is placed on a hot plate. Form a hypothesis about how the temperature will change over time. First, obtain water from the tap (ask Mr. Smith for help here) and fill up a 500 ml beaker with 300 ml of water. Then place the beaker on the hot plate along with the Venier temperature probe. Take note how the temperature changes. Every minute record the temperature and time. You will do this for 7 minutes. Once the time is up create a graph that shows how temperature changed over time.
| Graphing in Science Introduction | |
| File Size: | 2275 kb |
| File Type: | |
| Beach Pollution - 1 GIS | |
| File Size: | 1638 kb |
| File Type: | |
| Global Warming - 2 GIS | |
| File Size: | 3364 kb |
| File Type: | |
| Energy Resources - 3 GIS | |
| File Size: | 691 kb |
| File Type: | |
Properties of Water

In this lab you will examine the many properties of water. The properties of water include: adhesion, cohesion, low density when frozen and high heat capacity. Without these properties life on Earth as we know it would not be possible. Everything from plants to drinking straws to fish are effected by these properties.
You will go to five stations setup around the room performing the lab and answering the corresponding questions. Directions to each of the labs, as well as, followup questions can be found below.
You will go to five stations setup around the room performing the lab and answering the corresponding questions. Directions to each of the labs, as well as, followup questions can be found below.
Station 1: Cohesion
Directions:
Before you begin make a prediction of how many drops of water can fit on the penny. Using the eye dropper, carefully, place drops on the penny until the water spills off the penny.
Questions:
1.) How many drops fit on the penny?
2.) What did you observe about the "bubble" that forms on the top of the penny as you add more drops of water?
3.) How is water able to "stick" together?
Station 2: Adhesion
Directions:
Using the sharpie marker place a dot of ink about a 1/2 inch from the bottom. Place the paper towel into the water until the water level reaches the dot. Then allow the water to move up the paper towel. After about one minute take the paper towel out and measure the distance the water traveled up the paper towel.
Questions:
1.) How far up the paper towel did the water travel? (in centimeters)
2.) How does the water travel up the paper towel?
3.) How is adhesion on the paper compare to adhesion in plants?
Station 3: Adhesion
Directions:
Read the graduated cylinder to determine the amount of water within it. To read the amount you must look at the water level and notice that the water moves up the edge of the cylinder. This forms the "meniscus". You always read the water level by looking at the bottom of the meniscus.
Questions:
1.) How many milliliters are there in the graduated cylinder?
2.) How is the meniscus formed?
3.) Draw a picture of the meniscus.
Station 4: High Heat Capacity
Directions:
Observe the heat of the aquarium by placing your hand on the outside surface glass. Notice that inside the tank is a small thermostat that regulates the temperature for the fish.
Questions:
1.) How long do you think it takes to warm up this water? (Hint: is it very fast or slow?)
2.) How long does it take to warm up and cool down the oceans?
3.) How does this effect fish?
Station 5: Density
Directions:
Observe the beaker with ice and water.
Questions:
1.) How many physical forms of water are present?
2.) Where is the ice in the beaker? (Top, middle, bottom)
3.) Why does ice float?
4.) Which is more dense: water or ice?
Directions:
Before you begin make a prediction of how many drops of water can fit on the penny. Using the eye dropper, carefully, place drops on the penny until the water spills off the penny.
Questions:
1.) How many drops fit on the penny?
2.) What did you observe about the "bubble" that forms on the top of the penny as you add more drops of water?
3.) How is water able to "stick" together?
Station 2: Adhesion
Directions:
Using the sharpie marker place a dot of ink about a 1/2 inch from the bottom. Place the paper towel into the water until the water level reaches the dot. Then allow the water to move up the paper towel. After about one minute take the paper towel out and measure the distance the water traveled up the paper towel.
Questions:
1.) How far up the paper towel did the water travel? (in centimeters)
2.) How does the water travel up the paper towel?
3.) How is adhesion on the paper compare to adhesion in plants?
Station 3: Adhesion
Directions:
Read the graduated cylinder to determine the amount of water within it. To read the amount you must look at the water level and notice that the water moves up the edge of the cylinder. This forms the "meniscus". You always read the water level by looking at the bottom of the meniscus.
Questions:
1.) How many milliliters are there in the graduated cylinder?
2.) How is the meniscus formed?
3.) Draw a picture of the meniscus.
Station 4: High Heat Capacity
Directions:
Observe the heat of the aquarium by placing your hand on the outside surface glass. Notice that inside the tank is a small thermostat that regulates the temperature for the fish.
Questions:
1.) How long do you think it takes to warm up this water? (Hint: is it very fast or slow?)
2.) How long does it take to warm up and cool down the oceans?
3.) How does this effect fish?
Station 5: Density
Directions:
Observe the beaker with ice and water.
Questions:
1.) How many physical forms of water are present?
2.) Where is the ice in the beaker? (Top, middle, bottom)
3.) Why does ice float?
4.) Which is more dense: water or ice?
Macromolecules of Life: Tests for Organic Compounds

Understanding the chemistry of living organisms is an important part of biology. The structures of cells are made up of many different molecules. Cell metabolism involves the production and breakdown of many types of molecules. Most of the common molecules found in living things belong to four classes of carbon containing molecules: carbohydrates, lipids, proteins, nucleic acids.
Procedures:
Part A. Tests for Carbohydrates: Test for Starch
1.) Put on goggles and an apron. Label three test tubes 1,2,3. Place them in a test-tube rack.
2.) Using a separate dropper for each solution, add to each test-tube:
#1 -10 drops of starch solution
#2 -10 drops of glucose solution
#3 -10 drops of water
3.) Record the color of each tube's contents in Table 1.
4.) Add 3 drops of iodine to each test tube.
5.) Record in Table 1 the color of each tube's contents after addition of the iodine. A blue-black color indicates the presence of starch. Discard the contents of the test tubes according to Mr. Smith's direction.
Part B. Test for Lipids: Brown Paper test for Lipids
1.) Place a drop of water on a small piece of brown paper. Place a drop of oil on the same piece of paper. Allow the paper to dry for a few minutes.
2.) Hold the piece of paper up to the light. If a semi-transparent (translucent) spot is evident, the sample contains lipids.
3.) Throw away your paper towel and clean up your lab area.
Part B. Test for Lipids: Solubility test for Lipids
1.) Label two test tubes 1 and 2.
2.) Using separate droppers, add 20 drops of 95% ethanol to test tube 1 and 20 drops of water to test tube 2.
3.) Add 5 drops of oil to test tubes 1 and 2.
4.) Place your finger over the test tube and shake for about 20 seconds.
5.) Allow the contents of the tube to settle and record in Table 4 whether the oil is soluble (mixes together) in either solution.
*Lipids are soluble only in nonpolar solvents because lipids, themselves, are nonpolar. Water is polar; ethanol is not.*
6.) Dispose of the contents of the tubes according to Mr. Smith's instructions.
Part C. Tests for Proteins
1.) Label three test tubes 1, 2, 3.
2.) Using separate droppers, add to each test tube:
#1 - 30 drops of glucose
#2 - 30 drops of gelatin
#3 - 30 drops of water
3.) Record the color of the content in each test tube in Table 5.
4.) Add 10 drops of biuret reagent to each test tube.
*When biruet reagent is mixed with a protein, it will produce a purple to violet color.*
5.) Record in Table 5 the color of each tube's contents after adding biruet.
6.) Discard the contents of the test tubes according to Mr. Smith's instructions.
Venier Probe Investigation: pH
In this lab you will examine the pH of different liquids and identify a mystery substance. You will use Venier handheld devices to determine the pH of each substance.
To use the probe:
1.) Turn on the Venier handheld device (the power button is in the top left hand corner).
2.) Plug in the pH probe to the handheld device.
3.) Take the protective cover off the probe (be careful not to spill the buffer solution).
4.) Place the probe in water to wash it clean.
5.) Place the probe in the substance you wish to test and allow the probe time to measure the pH. The number will fluctuate, take the pH after about 15 seconds.
6.) After testing the substance, place the probe back in the water to wash it clean.
7.) Repeat steps 5 and 6 for each substance.
Identify the pH of each of the following substances:
Beaker A: Water pH:__________
Beaker B: Milk pH:__________
Beaker C: Coke pH:__________
Beaker D: Orange Juice pH:__________
Beaker E: Mystery Fluid pH:__________
Questions to answer:
1.) Which substance is the most acidic? Is this surprising to you? Why?
2.) Which substance is the most basic? Is this what you expected? Why?
3.) Are you surprised by the pH of water? Is it basic or acidic or neutral?
4.) Which beaker has the pH closet to the mystery beaker?
5.) What do you think the mystery fluid is?
6.) Draw a scale with all the pH of each fluid on it. (1-14)
<------------------------------------------------------------>
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14
To use the probe:
1.) Turn on the Venier handheld device (the power button is in the top left hand corner).
2.) Plug in the pH probe to the handheld device.
3.) Take the protective cover off the probe (be careful not to spill the buffer solution).
4.) Place the probe in water to wash it clean.
5.) Place the probe in the substance you wish to test and allow the probe time to measure the pH. The number will fluctuate, take the pH after about 15 seconds.
6.) After testing the substance, place the probe back in the water to wash it clean.
7.) Repeat steps 5 and 6 for each substance.
Identify the pH of each of the following substances:
Beaker A: Water pH:__________
Beaker B: Milk pH:__________
Beaker C: Coke pH:__________
Beaker D: Orange Juice pH:__________
Beaker E: Mystery Fluid pH:__________
Questions to answer:
1.) Which substance is the most acidic? Is this surprising to you? Why?
2.) Which substance is the most basic? Is this what you expected? Why?
3.) Are you surprised by the pH of water? Is it basic or acidic or neutral?
4.) Which beaker has the pH closet to the mystery beaker?
5.) What do you think the mystery fluid is?
6.) Draw a scale with all the pH of each fluid on it. (1-14)
<------------------------------------------------------------>
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14
Cell Interactive Game
In this online interactive game you will discover the different aspects of a cell's life. Click on the following link to go the the cell craft webpage.
http://www.carolina.com/category/teacher+resources/interactive+science+games+and+simulations/cellcraft.do
http://www.carolina.com/category/teacher+resources/interactive+science+games+and+simulations/cellcraft.do
Microscope Lab and Cell identification
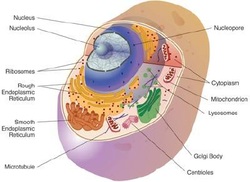
In this lab you will examine the multitude of cells that exist. You will examine each type of cell using a compound light microscope labeled #1-10. After examining each cell you will draw a picture and answer a question about the cell you view under the microscope.
Read this before you being the lab: How to use a microscope.
Station 1: Bird Ovary Cells
Draw an image of the cells.
What are the small pink circles within the ovary?
Station 2: Human Blood Cells
Examine this website: Blood Cells
Draw an image of the cells.
How many different type of cells do you see? (Hint: look for the different colors)
Station 3: Bird Intestine Cells
Draw an image of the cells.
Why is there a hole in the middle of the cells? What is it used for?
Station 4: Fish Cells
Draw an image of the cells.
What do you notice about the inside of each cell? What do you see?
Station 5: Plant Root Cells
Check out the different stages of each cell: Mitosis
Draw an image of the cells.
What shape do each of the cells appear to be in?
Station 6: Canary Plumage
Examine these examples of bird feathers.
Draw an image of the cells.
How does this image compare to the pheasant slide in #7?
Station 7: Pheasant Feather
Draw an image of the feathers.
How many total feathers are on the slide?
Station 8: Sperm Cells
Learn more about the form and function of sperm cells.
Draw an image of the cells.
What part of the sperm helps it move?
Station 9: Gold Fish Scale
Look at the many types of fish scales.
Draw an image of the cells.
How many scales are on the slide?
Station 10: Rainbow Trout Scale
Draw an image of the cells.
What differences do you notice between the gold fish and rainbow trout scales?
